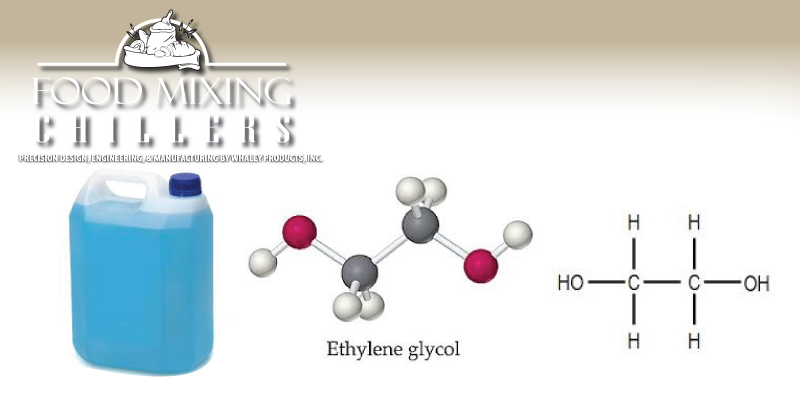
Ethylene glycol is a cooling agent used in chilling machines to make the product colder. Sometimes the glycol is contained in a metal jacket, or cycles through a network of pipes. A common name for ethylene glycol is antifreeze. Ethylene glycol is also used to de-ice runways for airports. Glycol is typically clear, miscible with water, odorless, and sweet tasting liquid. Miscible means can be easily mixed with water. Because of its sweet tasting nature, a bitter component is added so animals and humans do not consume it. Bright fluorescent dies are also added to indicate a leak in the flow of a system. Glycol is slightly viscous and breaks down in air in about 10 days. Ethylene glycol has little to no toxicity for long periods of time in low doses. But if a person ingests a large amount of the fluid, they can suffer dire consequences. The Central Nervous System will decline and a person will show symptoms such as coma, vomiting, seizures and renal damage. In some research involving small animals, liver damaged has been noted.